What if we fill a crater on the moon with water? Can we create a lake on the moon? Would the water stay in this artificial lake?
The short answer is “no”. The water would quickly freeze and then evaporate in short time. It would be a very short-lived lake.
Here is a beautiful explanation I found on Reddit:
We usually think of water as melting from ice to liquid water at 0 °C and boiling to steam at 100 °C. However, this is only true at atmospheric pressure. If we change the pressure, we get different melting and boiling points.
With this in mind, we can make a graph with temperature on the X-axis and pressure on the Y-axis and melting/boiling no longer being points, but being lines. We call this a “phase diagram”. Here is an approximate one for water below.
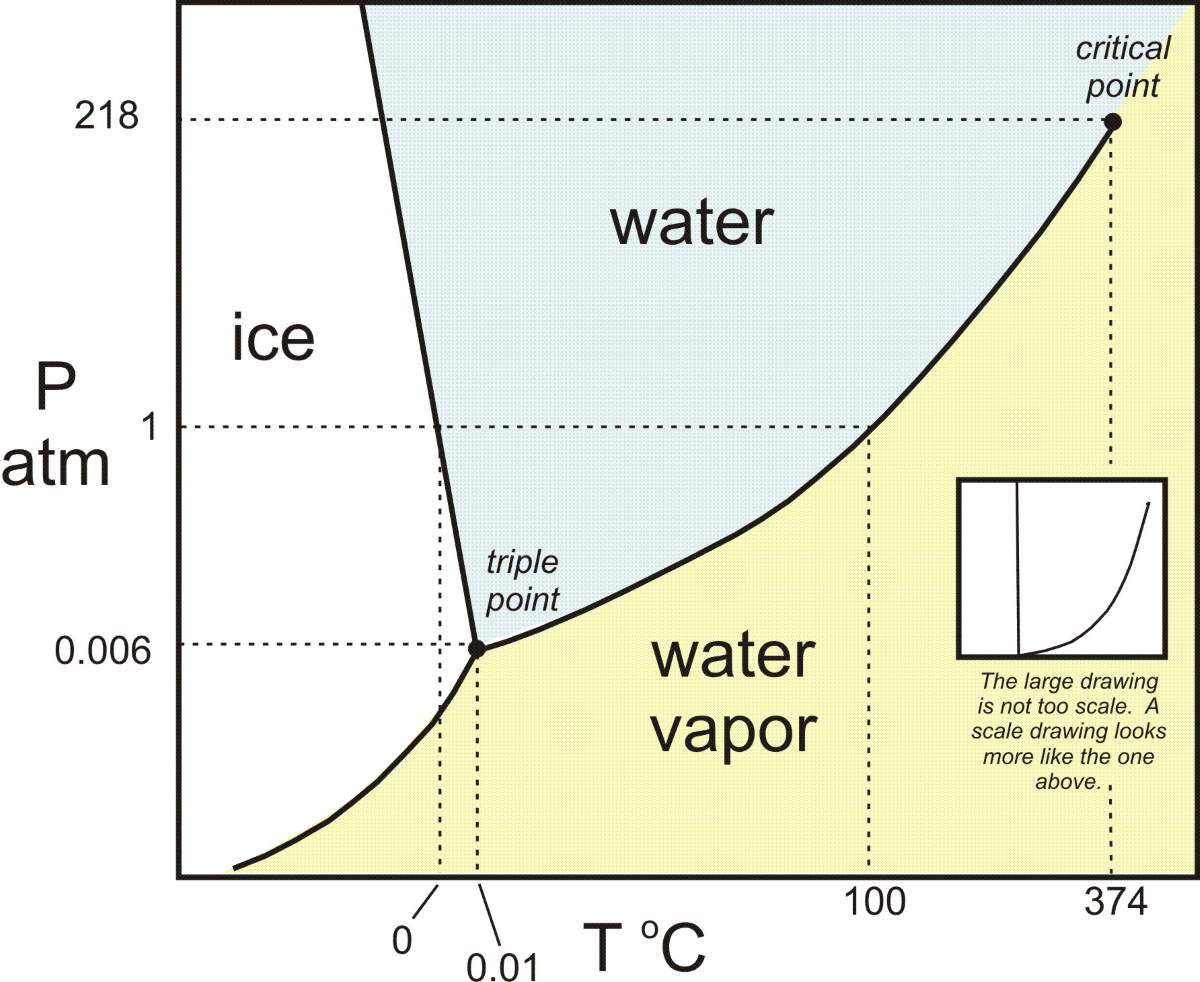
Moving along a horizontal line on the phase diagram is changing the temperature while at constant pressure while moving along a vertical line in the phase diagram is changing the pressure while at a constant temperature.
Notice that there is a point where the melting line (the line that separates the liquid and solid phases) and the boiling line (the line that separates the gaseous and liquid phases) meet. This is called the “triple point” because liquid, solid, and gas can all exist together at that temperature and pressure (see the video below).
Notice that there is another line below the triple point; one that separates solid from gas directly. This means that below the triple point’s pressure, there is no liquid phase and that once equilibrium is reached, only solid and gas will exist. The moon is, to a good approximation, a straight vacuum, so water can have no liquid state there at equilibrium.
If you were to make a lake magically appear on the moon without an atmosphere, it would immediately begin boiling. The boiling would carry away a lot of thermal energy (“heat”), which would cool off the remaining water until it was a solid block of ice. The solid ice would continue to sublimate (like melting but going directly from a solid to a gas without passing through a liquid phase) slowly until all the water was lost to a diffuse gas. Some ice underground or in perpetual shadow might persist, though.


Related: Amazing Moon facts
What is the “Critical Point” in the diagram?
In thermodynamics, a critical point (or critical state) is the endpoint of a phase equilibrium curve.
A ‘critical point’ occurs at the end of a phase line where the properties of the two phases become indistinguishable from each other, for example, when, under singular conditions of temperature and pressure, liquid water is hot enough, and gaseous water is under sufficient pressure that their densities are identical (0.322 g cm-3). At temperatures above the critical temperature, gas cannot be liquefied. Critical points are usually found at the high-temperature end of the liquid-gas phase line.
The existence of a critical point was first discovered by French engineer and physicist Charles Cagniard de la Tour (31 March 1777 – 5 July 1859) in 1822.
Russian chemist and inventor Dmitri Mendeleev (1834-1907) gave the name critical point” to this phenomenon in 1860.
Cagniard showed that carbon dioxide (CO2) could be liquefied at 31 °C at a pressure of 73 atm, but not at a slightly higher temperature, even under pressures as high as 3,000 atmospheres.
Sources
- Critical Point (thermodynamics) on Wikipedia
- Could you make a lake on the moon by filling a crater with water? Would the water stay in there? on Reddit
- Water Phase Diagram on lsbu.ac.uk
- How Many Elephants are Left in the World in 2025? - August 17, 2025
- Moon Landings: All-Time List [1966-2025] - February 2, 2025
- What Is Max-Q and Why Is It Important During Rocket Launches? - January 16, 2025